Personalized medicine is an evolving field of healthcare that is patient- and not anymore disease-oriented, in which physicians use diagnostic tests to determine the medical treatments that will work best for each patient.
More and more evidences indicate that many drugs are ineffective on some patients. For example, in 2001, drugs to cure depression were ineffective on 38% of patients, asthma drugs on 40%, diabetes drugs on 43%, arthritis drugs on 50%, Alzheimer’s drugs on 70% and cancer drugs on 75% of patients. Instead, by combining the data from diagnostic tests with an individual’s medical history, it is possible to develop targeted treatments and prevention plans. This has benefits for both patients and the healthcare system.
The first step to develop a personalized therapy is to identify the disease’s biomarkers, which is done through a genetic test. A biomarker is anything measurable that characterizes the diseases and distinguishes sick cells from healthy ones.
Biomarkers
Biomarkers are distinguished in four categories:
Predisposition biomarkers
A genetic predisposition (also called genetic susceptibility) is defined as the increased probability of developing a health disorder based on the presence of a particular biomarker. For example, a mutation in the BRCA1 and/or BRCA2 genes indicates that a patient has a higher risk to develop breast cancer. Usually, tests for genetic predisposition are done when someone has a personal or family history that suggests an inherited risk for a particular health condition, and when the results could provide information that help guide a person’s future medical care.
Diagnostic biomarkers
Diagnostic biomarkers are used to confirm that a patient has a particular disease. For example, the presence of mutations in the CFTR gene indicates that a newborn child has cystic fibrosis. Diagnostic biomarkers may facilitate earlier detection of a disorder than can be achieved by physical examination of a patient (PET, CT scan, X-rays and so on).
Prognostic biomarkers
A prognostic biomarker helps predict how a disease may develop in an individual, after diagnosis. The presence or absence of a prognostic marker can be useful for choosing the treatment but does not directly predict the response to it. For example, the diagnostic test Oncotype Dx examines 21 genes, to determine the likelihood that breast cancer will recur after initial treatment.
Predictive biomarkers
A predictive biomarker helps determine which patients are most likely to benefit from a specific treatment. Predictive diagnostics can provide information about how well a treatment is likely to work in a particular patient or about the likelihood of that treatment causing an unwanted side effect.
Personalized medicine
Personalized medicine aims to cure a disease depending on the biological mechanism that underlies it. In particular, this is relevant for tumors, which are caused by a broad spectrum of DNA mutations (tumor biomarkers). Even the same type of tumor can be caused by mutations in different genes. This heterogeneity results in different responses to therapies by tumors of the same kind, but with different genetic background. Now, thanks to biomarker-based diagnostic tests, it is often possible to identify the underlying mutation of a tumor, in order to plan an effective therapy against that specific biomarker.

One example of how personalized medicine is effective is given by a type of lung cancer, called non-small cell lung cancer. This type of cancer is usually caused by mutations in genes EGFR and/or ALK. Because these mutations are present only in the tumor cells, if the diagnostic test is positive for them, a specific therapy against these biomarkers can be chosen. This targeted therapy kills specifically tumor cells and not healthy ones (which do not have the biomarker), while traditional chemotherapy was not able to discriminate between the two and therefore gave heavy side effects.
Advantages of personalized medicine
First, thanks to predisposition biomarkers discovery, personalized medicine offers an opportunity to focus on prevention and early intervention rather than on reaction at advanced stages of the disease. As already mentioned above, women with mutations in BRCA1 or BRCA2 genes have up to an 85% lifetime chance of developing breast cancer, compared to a 13% chance among the general female population and also have up to a 39% and 17% chance, respectively, of developing ovarian cancer, compared to a 1,3% chance among the general female population. So, genetic tests for BRCA1 and BRCA2 genes can guide preventive measures, such as increased disease monitoring, chemo-prevention, or risk-reducing surgery.
Personalized medicine exploits drugs that target molecular characteristics of a disease. This often improves outcomes and reduces side effects and adverse reactions. About 30% of breast cancer cases are characterized by over-expression of a cell-surface protein called HER2. For patients with breast cancer that expresses this molecule, drugs that target HER2 can reduce their recurrence risk by 52%.
According to several studies, about 5,3% of all hospital admissions are associated with adverse drug reactions (ADRs). Often, ADRs are attributed to variations in genes that encode for drug-metabolizing enzymes. One example is a family of genes called cytochrome P450 (CYP450). Some variants of these genes cause drugs to be metabolized either faster or slower than normal. As a result, some individuals take longer time to inactivate and eliminate a drug from their body, leading to overexposure to it, while others eliminate the drug too rapidly before it has a chance to work. Personalized medicine takes this into account when selecting a drug for therapy and/or determining the dose.
A growing branch of personalized medicine includes gene- and cell-based therapies, which are designed to provide permanent or long-term benefits to patients by altering the molecular pathways associated with certain diseases. Gene- and cell-based therapies may involve replacing, repairing or inactivating a specific disease-causing gene or introducing a new/modified gene into a patient’s own cells to help treat a disease.
Another important advantage is the testing procedure, which often requires only a blood sample and therefore is much less invasive than tissue biopsies.
Last but not least, personalized medicine puts the focus on the patient: by integrating molecular diagnostic results with treatment decision-making, physicians can put aside the one-size-fits-all medicine and integrate patient perspectives in pursuit of shared clinical decision-making.
Ongoing trend
To identify biomarkers, molecular diagnostics requires DNA sequencing, which has a cost. Anyway, thanks to improving of sequencing technology, in the last years we observed a drastic decrease of costs, as displayed in the graph below.
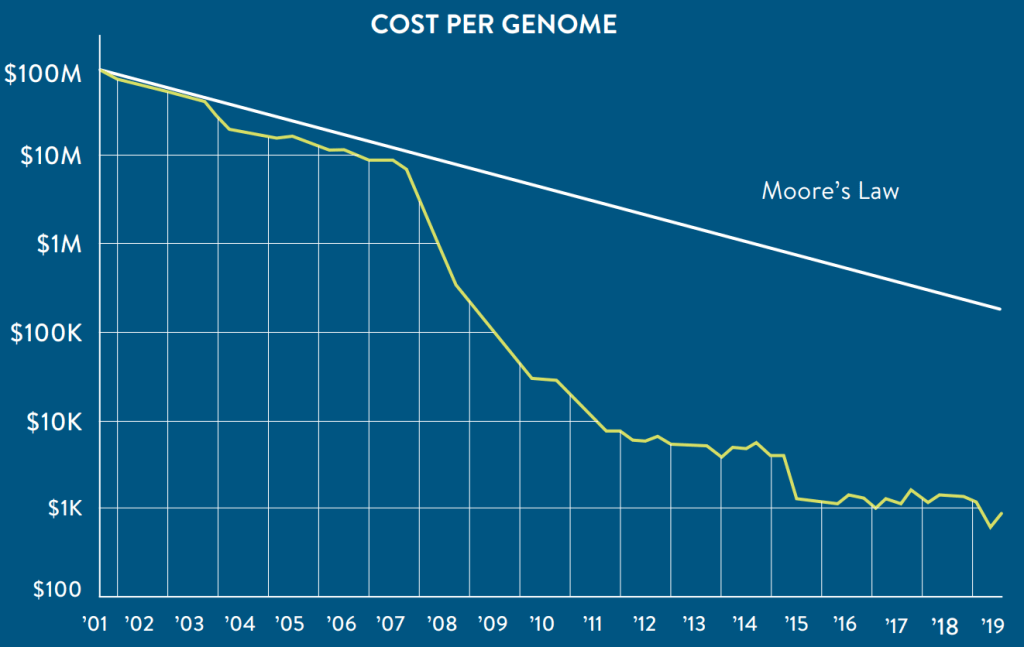
The higher accessibility to DNA sequencing technology leads to a constant increase in inclusion of biomarkers in clinical trials for cancer patients, as indicated in the picture below.

This resulted in a constant increase of personalized drugs approved for commercialization: in 2008 there were only 5, they became 81 in 2012, 132 in 2016 and in 2020 they are 286. Following the same trend, personalized medicines accounted for just 5% of the new drugs approved by the U.S. Food and Drug Administration (FDA) in 2005 and they became 25% in 2019.
The future
Personalized medicine is the future of healthcare. This implies several consequences:
- the requirement of novel ethical regulations (especially concerning gene therapy);
- a constant updating of physicians, who need to be aware of all the new treatment possibilities;
- healthcare delivery organizations must successfully integrate personalized medicine into clinical practice;
- health information systems must incorporate features that support 21st century medicine, providing the ability to collect and analyze clinical practice data and helping physicians make decisions based on the vast amount of information linking molecular patterns to diseases and treatment.
Policymakers must create an environment that encourages increased investment in diagnostics and targeted drugs, enables new advances in patient care that are safe, accurate and reliable and establishes a viable pathway toward patient access. In brief, healthcare need to follow the patient-centered strategy of personalized medicine, to get the best out of it.
