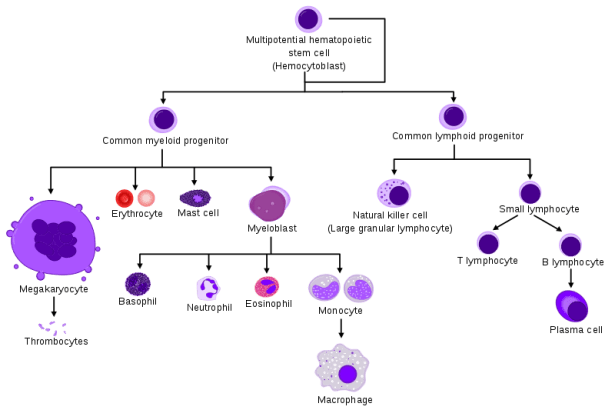
I write this post to make some clarification about hematological malignancies, because there are several kinds, with similar names but distinct features. Blood cancers are unique in their kind, because, differently than solid tissues, they are liquid and they circulate in the blood and in the lymphatic vessels. Hematopoietic diseases involve cells responsible for the immune response, at their different stages of maturation. Starting from a hematopoietic stem cell, two cell types differentiate (so-called lymphoid and myeloid progenitors). In turn, through a maturation process, they generate all hematopoietic cells. I will not go into details, but I want to show a picture (by Mikael Häggström and A. Rad) to give an idea of the complexity:
This said, let’s move on to the diseases. A first distinction is made between lymphoid and myeloid tumors, depending on the parental lineage of the mutated cell. Here I shortlist the few best-known diseases divided per lineage (tables are taken from “Overview of Hematologic Malignancies” written by Mikaela Olsen for Oncology Nursing Society), but I encourage you to check the references at the bottom of the post, to get a full picture.
Now you understand why I cannot enter details. Still, I will tell you something. In these tables, you read the word leukemia several times. The word derives from Greek words “leukos” and “heima”, which refer to excess of white cells in the blood. Leukemia was thought to be a single disease until 1877, when Paul Ehrlich invented a staining technique that allowed distinguishing the different cell types in the blood. This allowed a first classification of leukemia in chronic lymphocytic leukemia, chronic myeloid leukemia, acute lymphocytic leukemia, and acute myeloid leukemia, which became the reference classification for long time.
I explained here how tumors develop, and leukemia do not make exception. Mutations in the DNA are responsible for these cancers, and in particular, some translocations are peculiar for different types of leukemia. A translocation occurs when a piece of DNA from one chromosome moves and integrates in another chromosome. A translocation that occurs between chromosomes 9 and 22 generates the so-called chromosome Philadelphia, and is responsible for development of chronic myeloid leukemia. Translocation between chromosomes 15 and 17 is found in acute promyelocytic leukemia, instead. Different translocations cause different types of leukemia, because they alter the function of different genes. In other words, imagine that your car makes strange noises when you start it. It means that the engine has some problem. Nevertheless, which problem? If the noise is metallic, you might have an issue with valve tappets, if you hear some squeaking, probably the V-belt is worn out. In the same way, leukemia is caused by different DNA mutations, and the mutated gene(s), together with the cell type affected, determine its category.
What happens in leukemia? Tumor cells don’t die when needed, instead they continue proliferating and they create an unbalanced situation in the blood, where red blood cells find less room to deliver oxygen throughout the body. If the white blood cells proliferating abnormally are immature (closer to the stem cell parent), they proliferate fast and leukemia is acute; if they are mature, they proliferate at a slower rate, and the disease is chronic.
Together with leukemia, in the two tables you find often the word “lymphoma”. What’s the difference? In a gross way, we could say that leukemia is caused by mutations in the myeloid lineage, while lymphoma derives from lymphoid cells.
The myeloid lineage maturation occurs in the bone marrow, where in fact leukemia develops, causing an abnormal production of white blood cells. Lymphoma, instead, develops within the immune system sanctuary: the lymphatic system, where lymphocytes (that have lymphoid lineage) achieve complete maturation. Even though the lymphoid lineage starts maturation in the bone marrow, T-lymphocytes (cells that face directly the infection) become mature in the thymus. Other elements of the lymphatic system are lymph nodes, which filter lymph to modulate the immune response, and spleen, which hosts T- and B-lymphocytes (the cells responsible for antibodies production). Lymphoma is caused by mutations in lymphocytes. Among several types of lymphoma, the two main subclasses are Hodgkin (10-15% of the total) and non-Hodgkin lymphoma (85-90%). The former is specific for abnormal B-lymphocytes called Reed-Sterberg cells, while the latter can affect either T- or B-lymphocytes.
Now some numbers. How frequent are these liquid cancers?
An estimated combined total of 176,200 people in the US are expected to be diagnosed with leukemia, lymphoma or myeloma in 2019. 35% of these cases would be of leukemia, 47% of lymphoma and 18% of myeloma (another tumor that affects B-lymphocytes). Liquid tumors are expected to be the 10% of all tumors diagnosed in 2019 in US. 68,000 people are expected to die in US because of blood cancer.
In 2018, in the world, Non-Hodgkin lymphoma was the 11th most frequently diagnosed cancer (509,590 new cases, 3% of total cancer cases diagnosed), followed by leukemia at the 13th position (437,033 new cases, 2.6%), multiple myeloma (22nd, 160,000 new cases, 0.9%) and Hodgkin lymphoma (26th, 80,000 new cases, 0.5%).
SEER (Surveillance, Epidemiology and End Result) Cancer Statistics Review available in 2018 shows how the survival rate of these tumors increased along the years:

What about therapies?
Therapy for hematopoietic cancers follows two ways: bone marrow transplantation and precision medicine. The former is the gold standard that everyone heard of. For most of the diseases mentioned in the tables, it is the only possible therapy: to remove the cancerous bone marrow and replace it with a compatible and healthy one. But this not always works (e.g.: rejection, relapse).
Instead, for few diseases, it was possible to create a specific drug able to kill selectively tumor cells. In the case of chronic myeloid leukemia, translocation t(9;22), which causes the disease, generates a mutant protein that is not present in healthy cells. This is because the fusion point of the translocation fuses the gene BCR (normally present on chromosome 22) to the translocated gene ABL (normally on chromosome 9). This fusion protein BCR-ABL is a bold marker of tumor cells, making them easy to target with specific drugs. Unfortunately, situations like this one are difficult to happen, because usually mutations are smaller and difficult to detect. Therefore, in the majority of cases it is tough to design drugs specific enough to target only tumor cells, avoiding healthy ones.
References
Vardiman JW et al., The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood, 2009. Vol. 114, number 5. http://www.bloodjournal.org/content/bloodjournal/114/5/937.full.pdf?sso-checked=true
Swerdlow SH et al., The 2016 revision of the World Health Organization classification of
lymphoid neoplasms. Blood, 2016. Vol. 127, number 20. http://www.bloodjournal.org/content/bloodjournal/127/20/2375.full.pdf?sso-checked=true
Olsen M. Overview of Hematologic Malignancies. Nursing Cancer Society. https://www.ons.org/sites/default/files/publication_pdfs/HM%20Ch%20%201.pdf
Mikael Häggström and A. Rad – Image: Hematopoiesis (human) diagram.png by A. Rad, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=7351905
Facts 2018-2019 provides updates from the American Cancer Society’s Cancer Facts & Figures 2019 (published online in 2019, https://www.cancer.org/research/cancer-facts-statistics.html) for estimated numbers of new blood cancer cases and estimated numbers of deaths due to blood cancers.
The incidence rates, prevalence and mortality data in Facts 2018-2019 reflect the statistics from the National Cancer Institute’s Surveillance, Epidemiology and End Results (SEER) Program, Cancer Statistics Review (CSR) 1975-2015 (published online in April 2018, http://www.seer.cancer.gov).
https://www.wcrf.org/dietandcancer/cancer-trends/worldwide-cancer-data


